Lab-on-a-chip synthesis of cosmic dust analogues
Middle/Upper Atmosphere Laboratory, Past Research.
Leverhulme Trust Award 2011
The aims of this project were to develop synthetic analogs for cosmic dust or meteoric material, to be used in investigating a number of gas/solid processes of interest in the atmospheres of Venus, Earth, Mars and Titan (and exoplanets with similar atmospheres).
Two synthesis methods have been employed;
- Bulk synthesis (up to 1 L volumes) from appropriate precursors. This produces significant mass (several grams) of relevant particles in a reasonably short period of time, and at very low cost. Dialysis using a soxhlet apparatus has been employed to remove precursors and byproducts.
- Lab on a chip synthesis. This is described by the scheme shown below. Small amounts of particles are produced, but in a much more environmentally relevant size range.
These particles have been characterised by:
- Dynamic Light Scattering (DLS) - Measures size distributions
- X-Ray Diffraction (XRD) - Crystal structure determination
- Scanning Electron Microscopy (SEM) - Micron scale morphology
- Transmission Electron Microscopy (TEM) - Nanometer scale morphology
- Energy Dispersive X-Ray Spectroscopy (EDX) - Elemental composition determination (during both TEM and SEM analysis)
- Brunauer, Emmett and Teller Isothermal Adsorption (BET) - Available surface area
- Ellipsometry / Spectroscopy - Measurement of refractive indexes
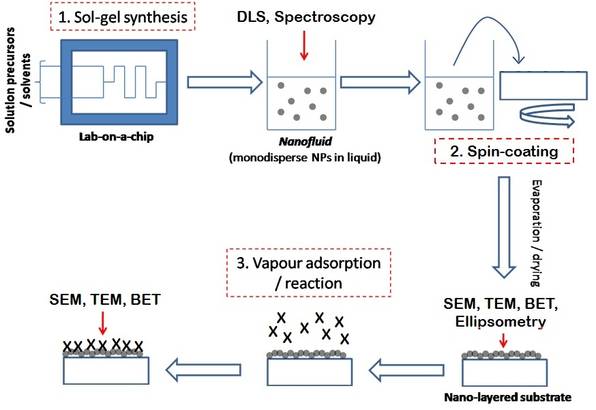 Operating scheme for the Lab on a Chip apparatus.
Operating scheme for the Lab on a Chip apparatus.
HNO3 uptake on Meteoric Smoke
As an example of the applications to which synthetic meteor smoke has been applied, uptake coefficients have been measured for nitric acid on synthetic amorphous olivine. This is thought to be an important destruction pathway for nitric acid produced by auroral precipitation. Our experiments, coupled with atmospheric modelling of meteoric smoke particles, show that this is the dominant destruction pathway during the polar winter. The figure below shows the modeled impact of varying the uptake coefficient (γ) on the percentage of HNO3 which the MSP would remove.
